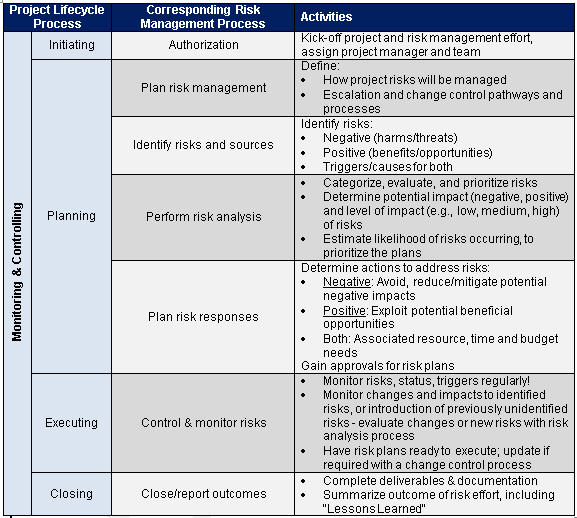
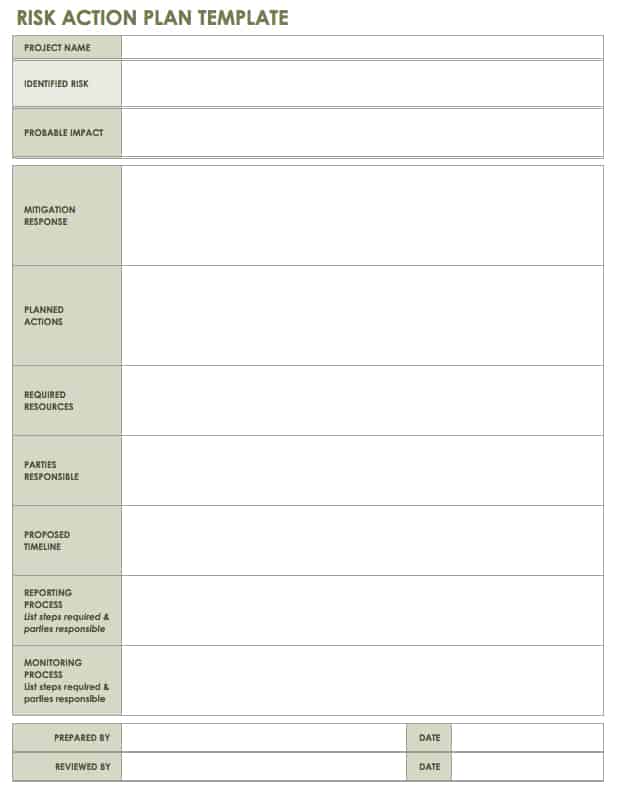
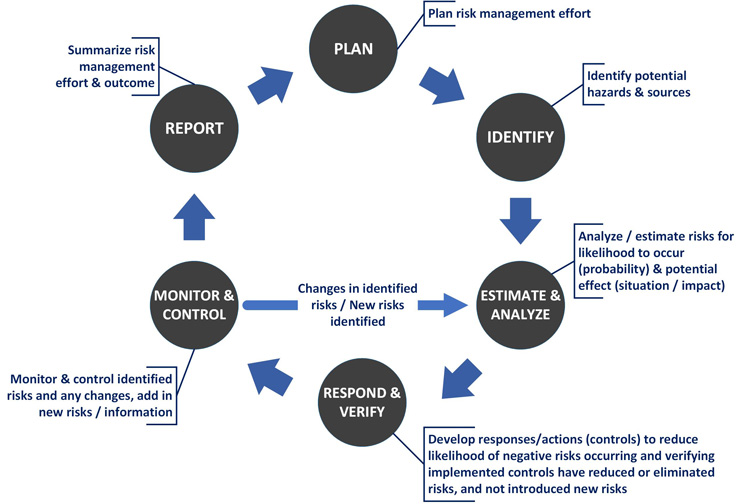
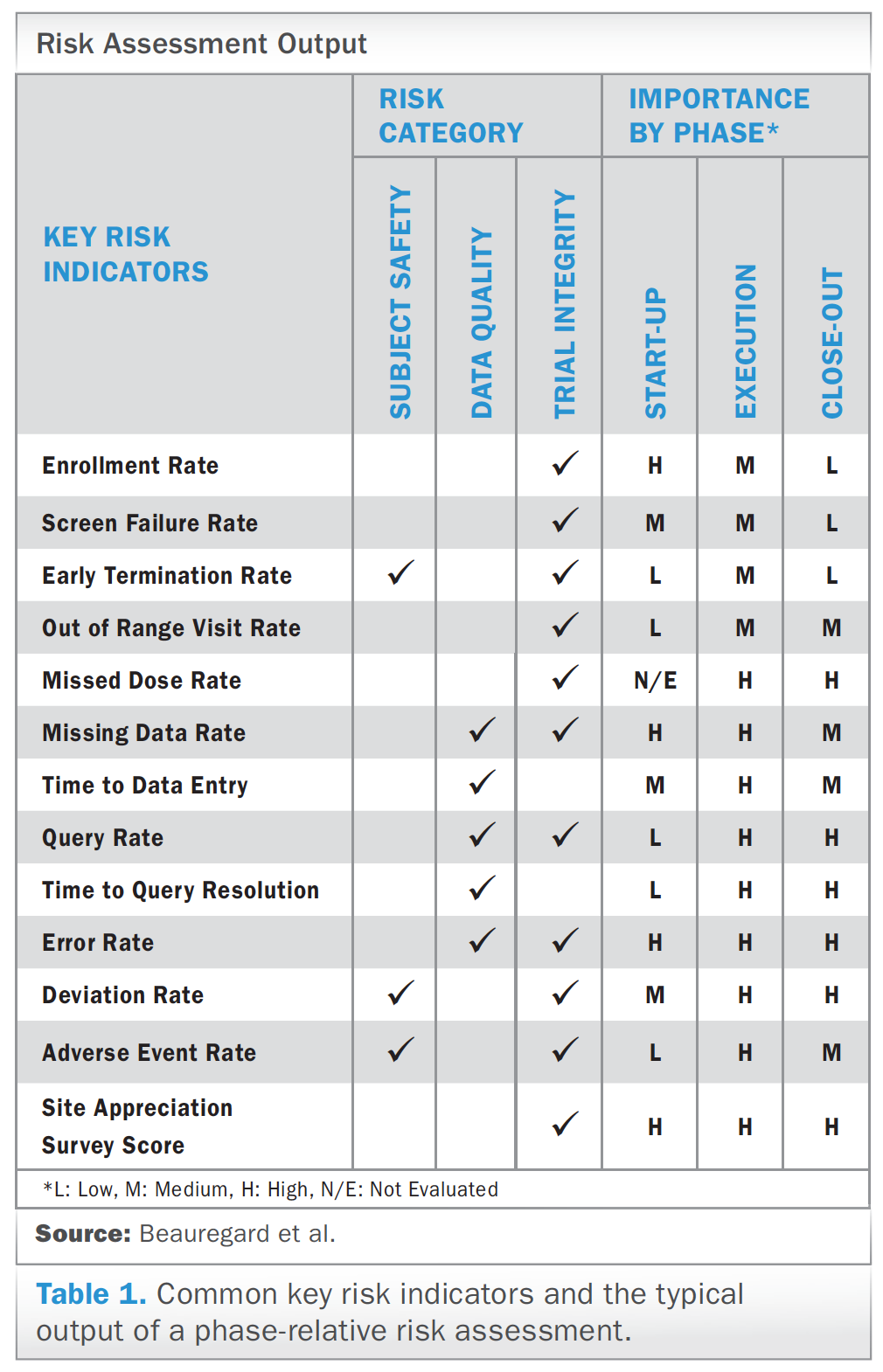
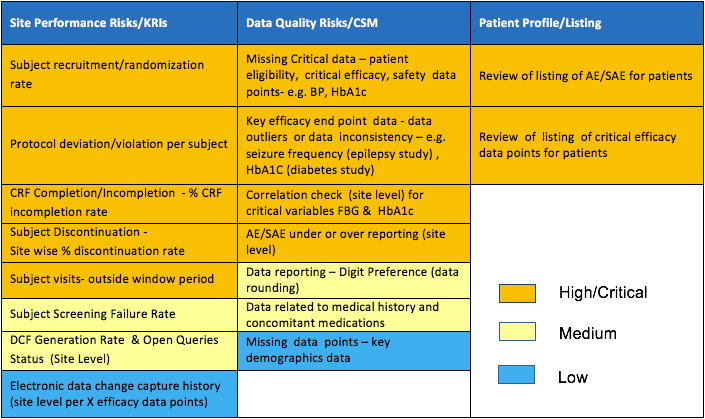
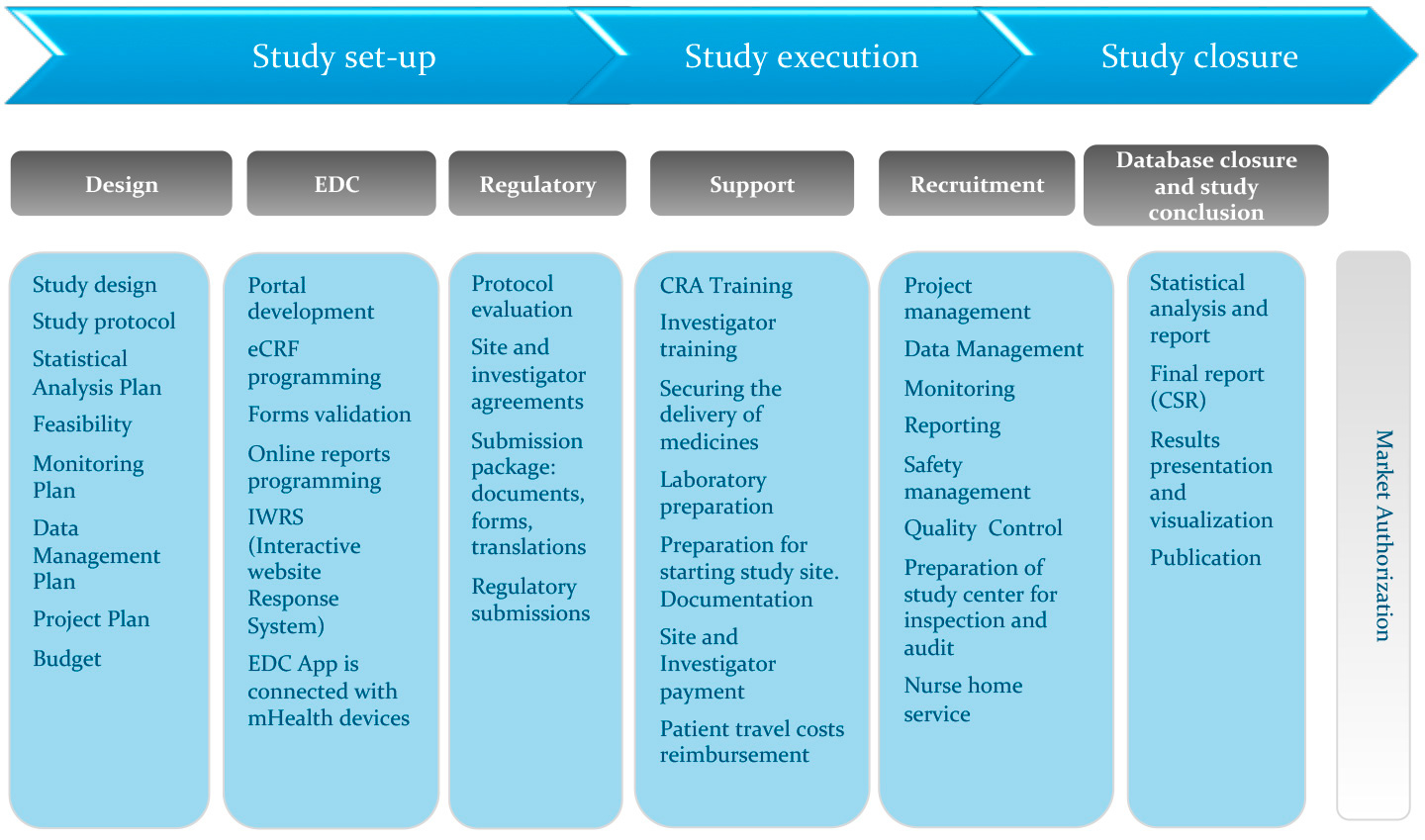
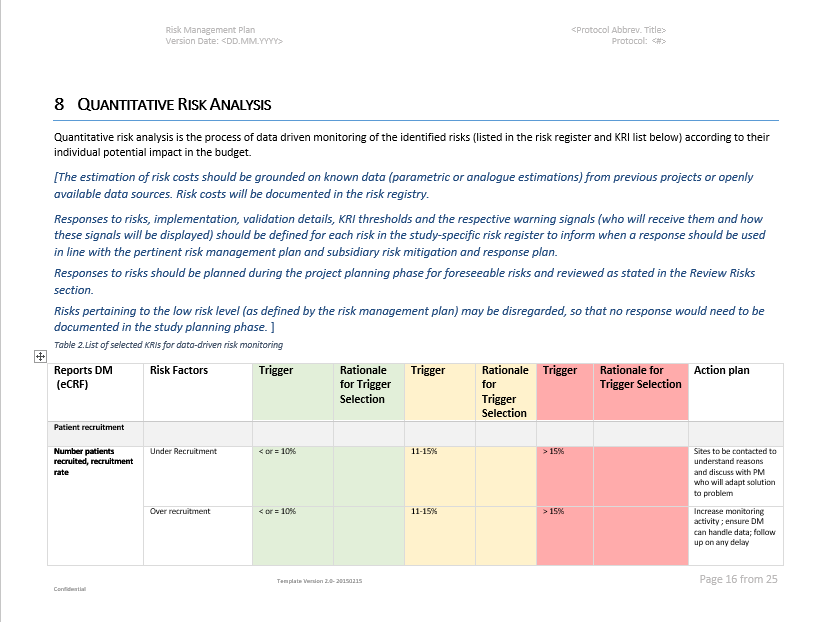
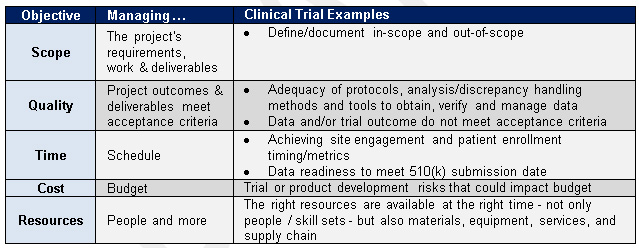
Risk Management – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India

Safety Management Plan – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India
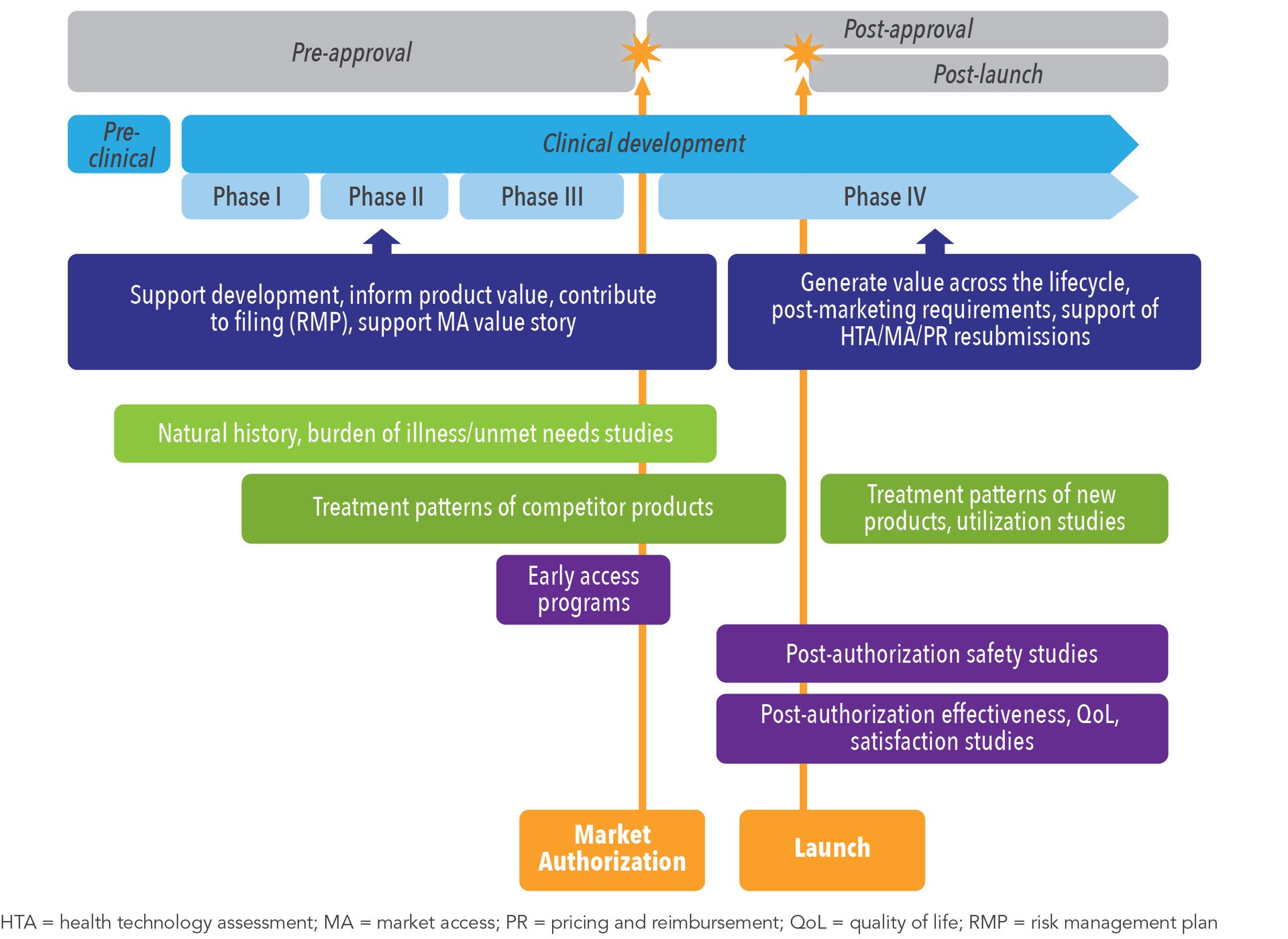
White Paper: Protocol Design in Real-World Evidence: The Indispensable Link Between Strategic Need and Study Execution - Evidera