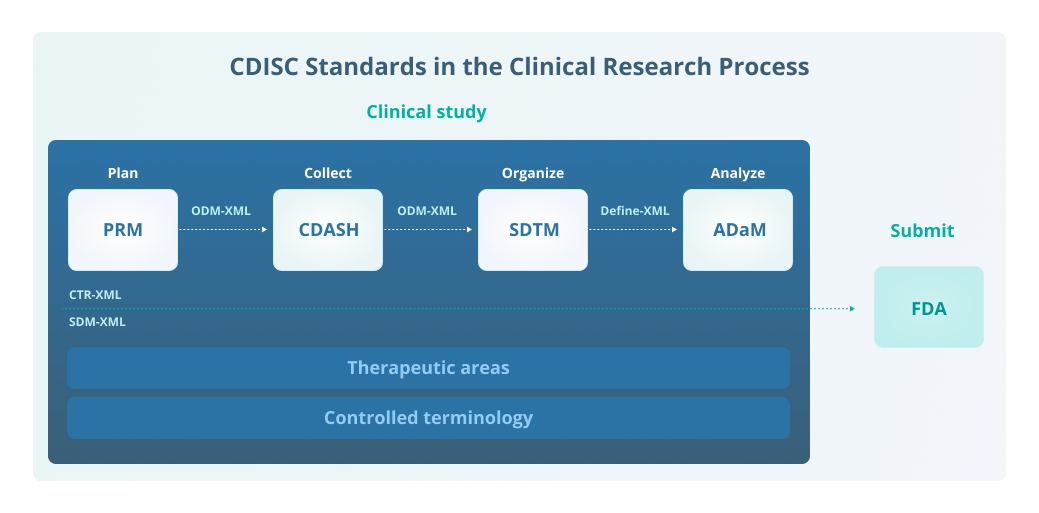
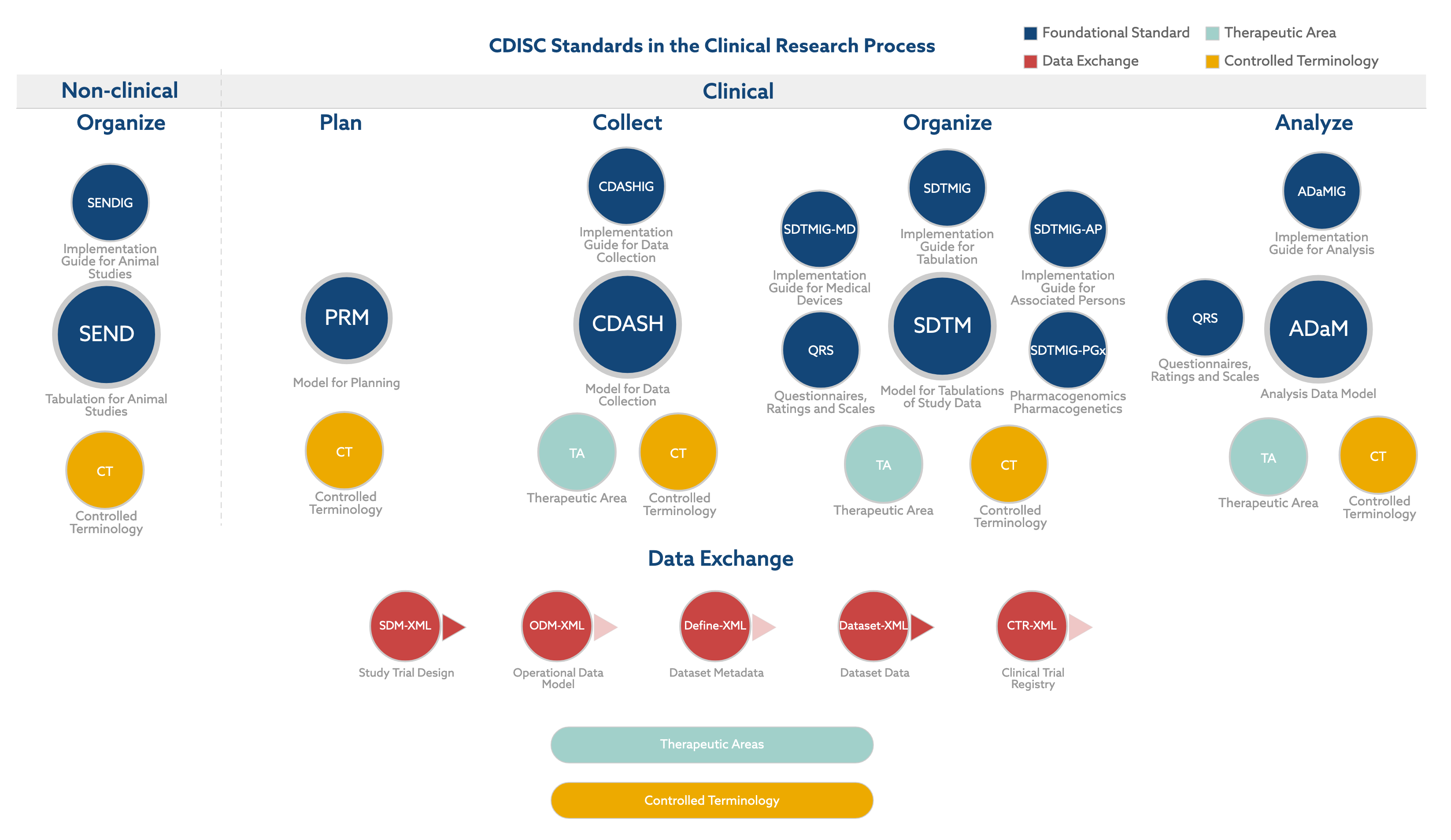
![PDF] CDISC Transformer: a metadata-based transformation tool for clinical trial and research data into CDISC standards | Semantic Scholar PDF] CDISC Transformer: a metadata-based transformation tool for clinical trial and research data into CDISC standards | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/39c5a0f62852e66837394ef1953006c33a1d8587/6-Figure3-1.png)
PDF] CDISC Transformer: a metadata-based transformation tool for clinical trial and research data into CDISC standards | Semantic Scholar
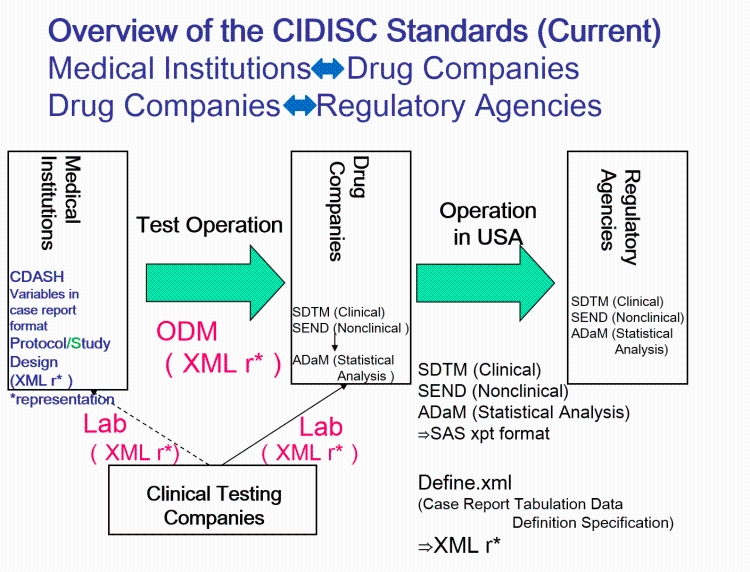
Joining the Clinical Data Interchange Standards Consortium (CDISC) and Activity Status | Japan Agency for Medical Research and Development

OpenClinica becomes Gold Member with the Clinical Data Interchange Standards Consortium (CDISC) » OpenClinica

Sections of CDISC ODM with study metadata information left-hand side... | Download Scientific Diagram

A pragmatic method for transforming clinical research data from the research electronic data capture “REDCap” to Clinical Data Interchange Standards Consortium (CDISC) Study Data Tabulation Model (SDTM): Development and evaluation of REDCap2SDTM -