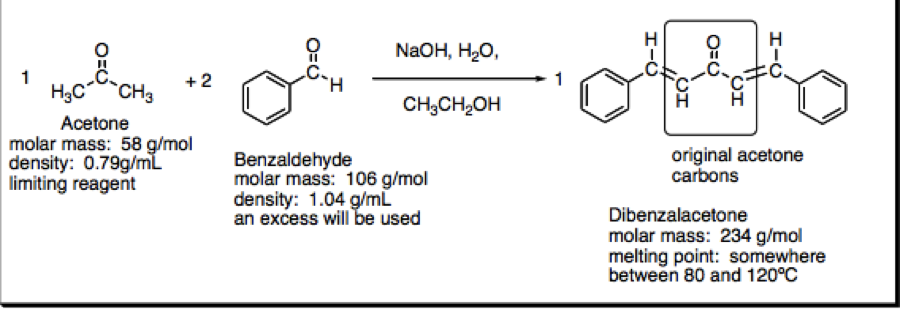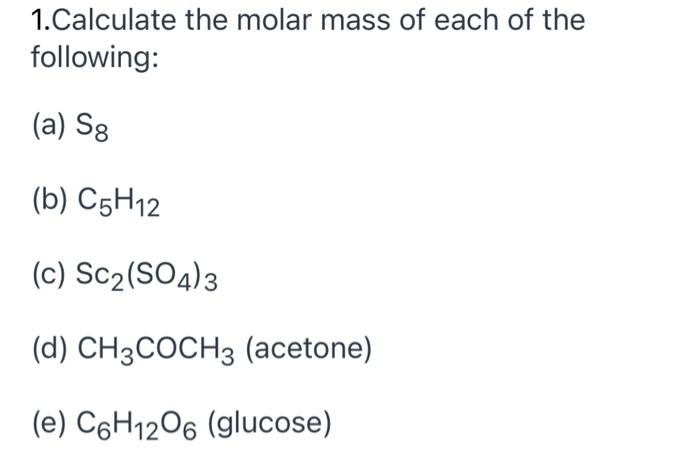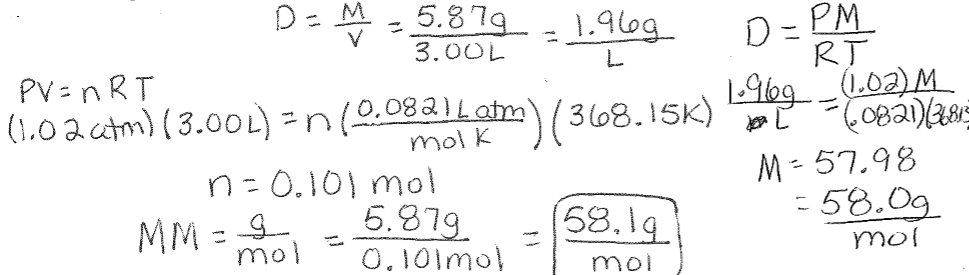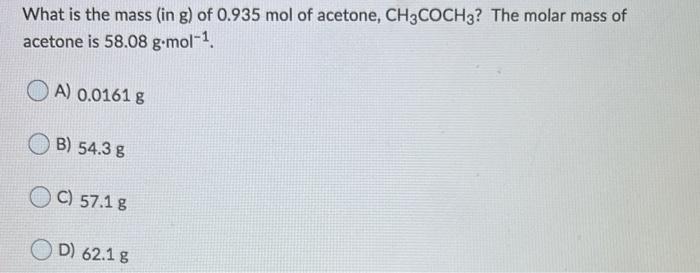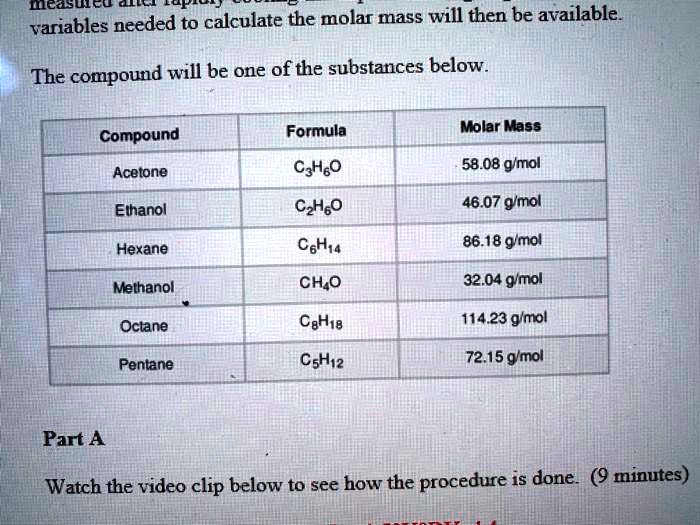
SOLVED: Mcdsucu 4 variables needed to calculate the molar mass will then be available: The compound will be one of the substances below Formula Molar Mass Compound Acetone CyHso 58.08 gmol Ethanol
![Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube](https://i.ytimg.com/vi/CCWl-z3Wdmo/sddefault.jpg)
Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube

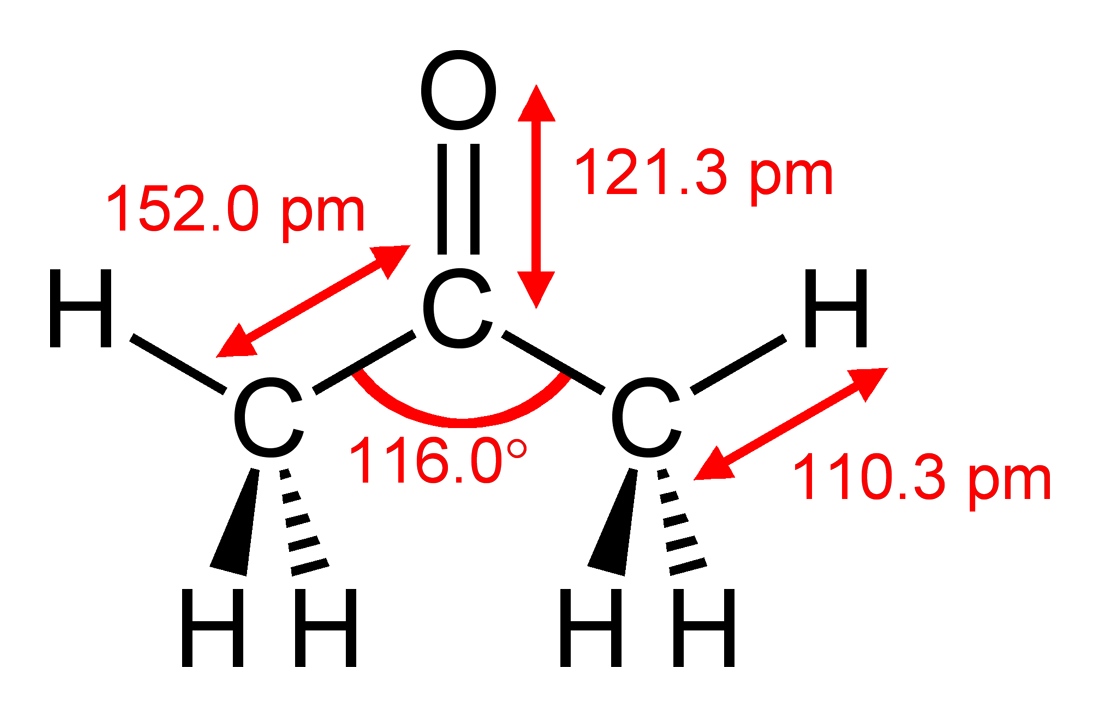
Acetone Molecule Structure & Chemical Formula | What is Acetone? - Video & Lesson Transcript | Study.com
Solved] Calculate the molar mass of each of the following: Sc2(SO4)3 CH3COCH3(acetone) C6H12O6(glucose) Calculate the empirical or molecular formula... | Course Hero

5.8 gram of acetone (CH3COCH3)is dissolve in 90 gram water, calculate mole fraction of water and - Brainly.in

Sample of mixture of A(g), B(g) and C(g) under equilibrium has a molecular weight (observed) is 80.The equilibrium is A(g) B(g) + C(g) (mol. wt. = 100) (mol. wt. = 60) (mol.

A student heats a beaker containing ice and water. He measures the temperature of the contents of the beaker as a function of time.Which of the following would correctly represent the result?

Please solve the 10th question I'm not able to understand why in the solution the latent heat per mole - Chemistry - Solutions - 16374635 | Meritnation.com

Size of PCL (molar mass M w = 25 kg/mol) NPs prepared from an acetone... | Download Scientific Diagram

SOLVED: A chemist wants to determine the molar mass of a naturally occurring substance isolated from spearmint. She adds 4.531 g of this substance to 25.00 g of acetone (C3H6O; molar mass